Context
The laboratory is focused on developmental biology of plants using the model system Arabidopsis thaliana. The aim of this study was to investigate the intracellular localization of protein receptors (CLV1, CLV2, and CRN) in plant cells and their tendencies for protein-protein interactions. To analyze receptor localization and interaction, a transient expression system in Nicotiana benthamiana leaf epidermis cells was used [1].
[1] A. Bleckmann et al., Plant Physiology, January loading control. wt, wild type. 2010, Vol. 152, pp. 166–176, www.plantphysiol.org
Materials
- Precellys®24homogenizer.
- Precellys® kit: 03961-1-003 (ceramic beads 1.4mm)
- Sample : ~0.1 g of N. benthamiana leaf tissue.
- Extraction buffer: 750 µL (0.1 M Tris-HCl, pH 8.3, 5 mM dithiothreitol, 5 mM EDTA and protease inhibitor).
Protocol
- Plant tissue homogenized using the Precellys®24: 5500 rpm, 1×20 sec.
- 1h incubation of plant extract at 4°C followed by 10 min denaturation at 95°C.
- Protein separation by SDS-PAGE & Western-blot analysis.
Results
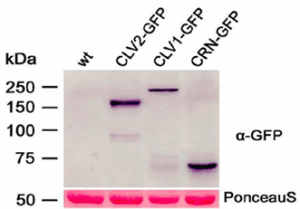
Our transient expression studies of translational fusions with GFP or mCherry now showed that all three receptor proteins can localize to the plasma membrane and have the capacity to undergo multiple interactions. The Precellys®24 is a fast method for protein isolation. Protein extracts were used to demonstrate fusion protein expression and stability.
Figure 1: Western-blot analysis of protein extracts from N. benthamiana leaf cells transiently expressing CLV1-GFP, CLV2-GFP, or CRN-GFP. An anti-GFP antibody was used for detection; sizes of protein markers are given in kD. The Ponceau Sstained protein bands of Rubisco are shown as a loading control. wt, wild type